|
11/21/2023 0 Comments Sigma bond shape 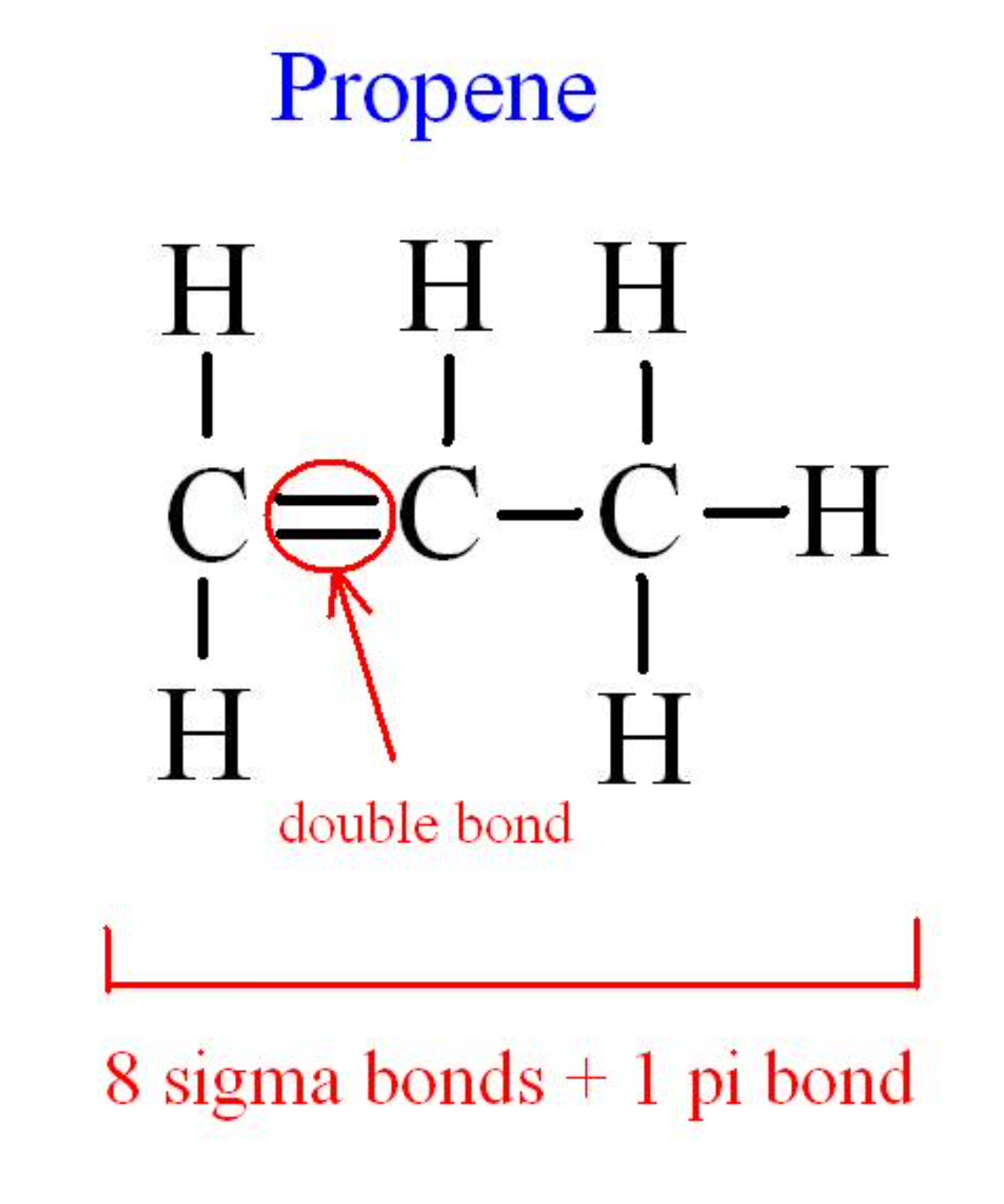 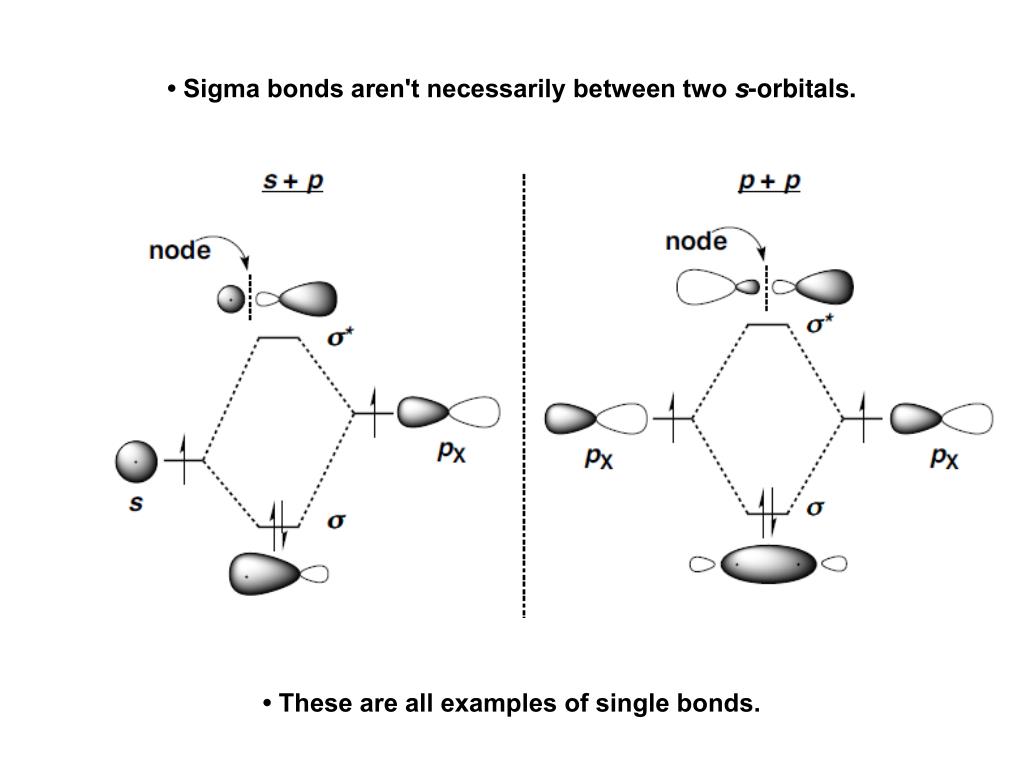
The valence shell electron pair repulsion (VSEPR) model focuses on the bonding and nonbonding electron pairs present in the outermost (valence) shell of an atom that connects with two or more other atoms.įundamentally, the VSEPR model theorizes that these regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Experimentally, the four carbon-hydrogen bonds in the ethylene molecule have been shown to be identical. The Lewis structure of ethylene indicates that there are one carbon-carbon double bond and four carbon-hydrogen single bonds. Sigma bonds are made by the overlap of two hybrid orbitals or the overlap of a hybrid orbital and a s orbital from hydrogen. Ethylene (commonly knows as ethene), CH 2 CH 2, is the simplest molecule which contains a carbon carbon double bond. A sigma bond can be formed by overlap of an s atomic orbital with a p atomic orbital. A triple bond is made up of a sigma bond and two pi bonds. This type of bond is referred to as a (sigma) bond. VSEPR Theory: a chemistry model used to predict the shape of individual molecules based on electron-pair electrostatic repulsion A double bond is made up of a sigma bond and a pi bond.The main geometries without lone pair electrons are: linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral.Molecular geometries take into account the number of atoms and the number of lone pair electrons. In sp³ hybridization, one s orbital and three p orbitals hybridize to form four sp³ orbitals, each consisting of 25 s character and 75 p character.Fundamentally, the VSEPR model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
